Drug Effect Comparison Tool
Understand Your Medication's Side Effects
This tool compares on-target versus off-target side effect profiles based on the type of drug you're taking. Select a drug class to see real-world side effect statistics from the article.
On-Target Effects
PredictableSide effects that occur because the drug is working as intended, but in tissues where it's not wanted.
Example
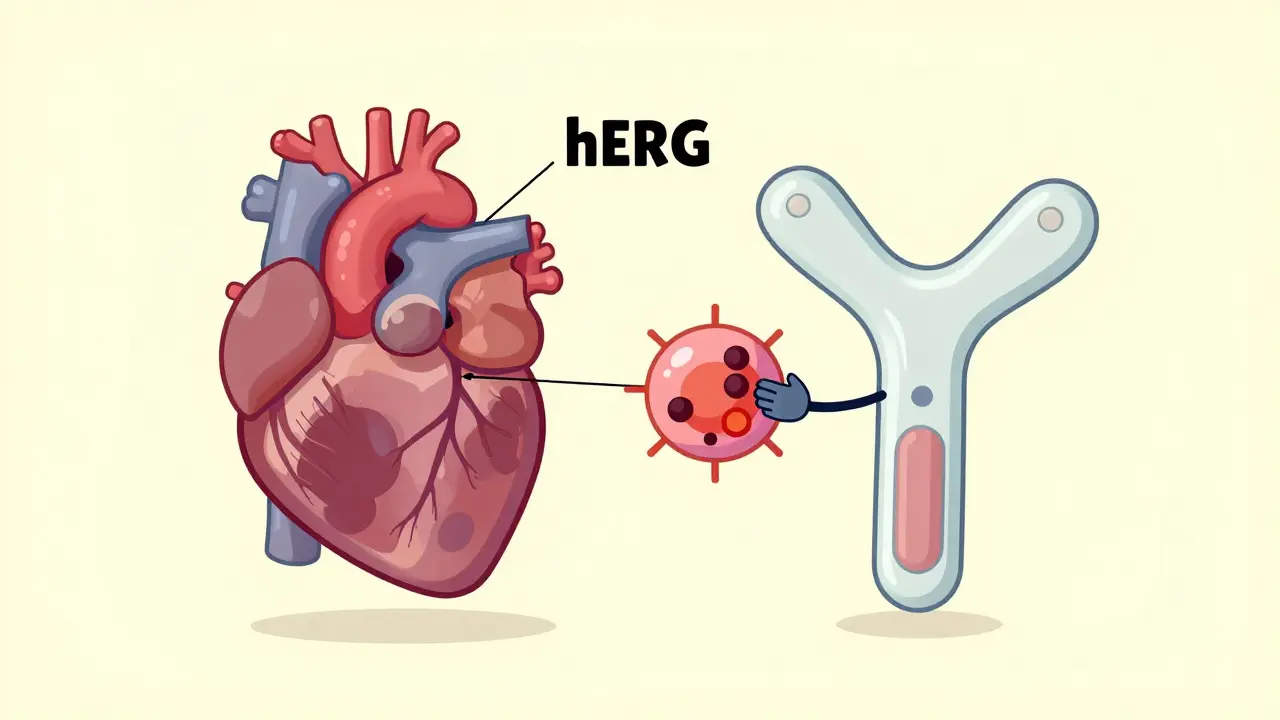
EGFR inhibitors causing skin rash
Frequency
68%
Off-Target Effects
UnpredictableSide effects that occur when the drug binds to unintended targets in the body.
Example
Thalidomide causing birth defects
Frequency
Rare but dangerous
Key Differences
Small Molecule Drugs
Average 6.3 off-target interactions at therapeutic doses
High risk of unexpected side effects
Biologics
Average 1.2 off-target interactions
Lower risk of off-target effects
Have you ever taken a medication and wondered why you got a side effect that seemed totally unrelated to what the drug was supposed to do? Maybe your diabetes pill gave you stomach cramps, or your blood pressure med made your skin itch. It’s not random. There’s a science behind why drugs cause side effects-and it all comes down to two things: on-target and off-target effects.
What Exactly Is an On-Target Effect?
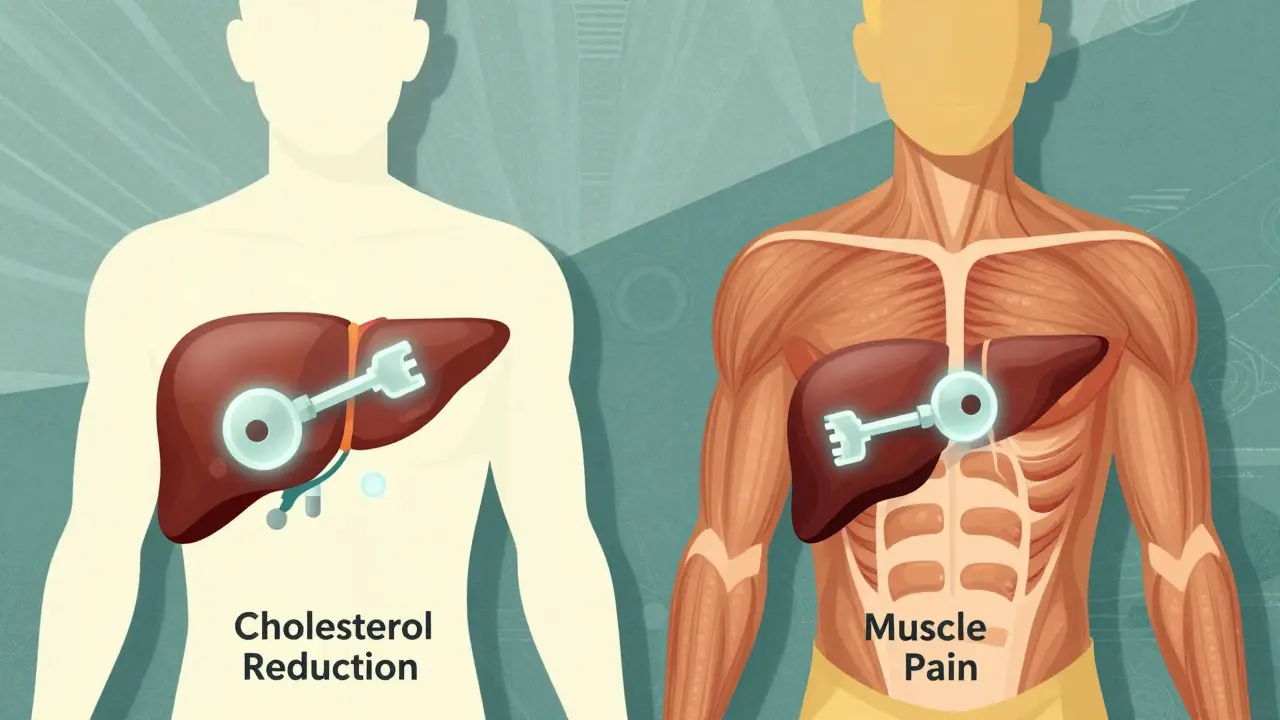
An on-target effect is when a drug does exactly what it’s designed to do-but in the wrong place. Think of it like a key that fits perfectly in one lock (the target), but accidentally turns that same lock in other rooms. For example, statins lower cholesterol by blocking HMG-CoA reductase, an enzyme in the liver. That’s the on-target effect. But if that same enzyme is also active in muscle cells, blocking it there can cause muscle pain or even rhabdomyolysis. That’s not a mistake-it’s still the drug working as intended, just in a tissue it shouldn’t be affecting.On-target side effects are common in cancer drugs. EGFR inhibitors, used for lung cancer, often cause severe skin rashes because the EGFR protein is also critical for healthy skin cell turnover. MEK inhibitors can lead to vision problems because they interfere with signaling in the retina. These aren’t accidents. They’re predictable, dose-dependent, and often manageable. In fact, 68% of patients on EGFR inhibitors develop skin issues, and doctors expect them. They adjust doses or prescribe topical creams. These side effects are frustrating, but they’re not a surprise to clinicians.
What Makes Off-Target Effects So Dangerous?
Off-target effects are the real wild cards. These happen when a drug binds to something it wasn’t meant to. A drug designed to block a cancer protein might accidentally latch onto a heart ion channel, a liver enzyme, or even a receptor in the gut. The result? Toxicity that’s hard to predict, often rare, and sometimes deadly.Take thalidomide. Originally developed as a sleep aid and anti-nausea drug for pregnant women, it caused horrific birth defects because it interfered with blood vessel formation in developing limbs. That wasn’t its target. It was an off-target effect. Fast-forward decades, and now thalidomide is used to treat multiple myeloma-because its off-target immune-modulating properties turned out to be useful. This flip-flop shows how off-target effects can be both dangerous and, sometimes, therapeutic.
Small molecule drugs are especially prone to off-target interactions. Studies show they bind to an average of 6.3 unintended targets at therapeutic doses. Kinase inhibitors, like imatinib (Gleevec), are notorious. They were designed to block BCR-ABL in leukemia, but they also hit c-KIT and PDGFR. That’s why they work against gastrointestinal stromal tumors-but also cause fluid retention and swelling. In contrast, monoclonal antibodies like trastuzumab (Herceptin) are more selective. They’re large proteins that bind tightly to one specific target, so they have far fewer off-target effects.
Why Some Drugs Fail-and Others Succeed
About 40% of drug candidates fail in Phase II clinical trials because of unexpected toxicity. Of those failures, 65% are due to off-target effects. That’s why big pharma spends millions on screening tools before a single human trial begins.Companies like Genentech and Novartis use technologies like KinomeScan to test how a drug interacts with hundreds of proteins at once. They don’t just look at the intended target-they scan the whole cellular landscape. This helps them spot risky interactions early. A drug that binds to 20 different kinases? It’s probably doomed. One that sticks cleanly to just one? It has a much better shot at approval.
There’s a financial incentive too. Drugs with clean target profiles-minimal off-target effects-generate 34% more revenue over their patent life. Why? Because patients stay on them longer. Doctors prescribe them more confidently. Pharmacies stock them more readily. And regulators approve them faster.
The Hidden Complexity: It’s Not Just About the Target
Here’s the twist: even when a drug hits the right target, the effect isn’t always the same. A 2019 study in Nature Scientific Reports looked at four different statins across three cell types. At the gene level, each statin triggered completely different changes. But when researchers looked at pathways-groups of genes working together-they found consistent patterns. All statins activated the same cholesterol-lowering pathway. But immune-related pathways? Those varied wildly. That means: the same drug can behave differently depending on the cell type.This explains why some people get side effects and others don’t. Two patients on the same dose of a blood pressure drug might react differently because their liver enzymes, gut microbiome, or genetic background alter how the drug is processed-or which off-target proteins it binds to.
Real-World Examples You Can Relate To
- Metformin: Used for type 2 diabetes, it lowers blood sugar by reducing glucose production in the liver (on-target). But it also acts on gut bacteria and intestinal cells, causing diarrhea and bloating. That’s still an on-target effect-just happening in the gut instead of the liver. Patients often think it’s an allergy. It’s not. It’s the drug working too well in the wrong place. - Sildenafil (Viagra): Originally developed for angina, it was meant to relax blood vessels in the heart. But researchers noticed it also caused penile erections. That wasn’t the goal. It was an off-target effect. Now, it’s one of the best-selling drugs in the world. - Chloroquine: Used during early COVID-19 trials, it was thought to block viral entry. But later studies showed it didn’t bind to the virus-it disrupted acid levels in cellular compartments like lysosomes. That’s an off-target effect. It didn’t work for COVID, and it caused dangerous heart rhythms in some patients.
What’s Changing in Drug Development?
The old way of drug discovery was simple: find a target, make a drug, test it. Now, it’s systems-level. Companies don’t just look at one protein. They look at gene expression, protein networks, metabolic changes, and even how the drug affects RNA. Tools like the Open Targets Platform combine genomic data, chemical structures, and clinical outcomes to predict off-target risks before a drug is even made.Regulators are catching up. The FDA and EMA now require at least two independent methods to check for off-target effects in gene therapies and new small molecules. AI models trained on millions of drug interactions can now predict off-target binding with 87% accuracy. AstraZeneca cut off-target toxicity predictions by 42% just by adding metabolomic data to their screening pipeline.
What Does This Mean for You?
If you’re a patient: side effects aren’t always mistakes. Some are expected. Others are lucky breaks. If your drug causes a weird symptom, don’t assume it’s an allergy. Talk to your doctor. Ask: Is this on-target or off-target? That distinction can change how you manage it.If you’re a student, researcher, or just curious: understanding this difference is the key to modern medicine. The future of drug development isn’t about making drugs that hit one target perfectly. It’s about making drugs that hit the right target-and avoid the wrong ones.
And here’s the bottom line: the best drugs aren’t the most specific ones. They’re the ones that strike the right balance-effective enough to work, clean enough to be safe.
Are all side effects caused by off-target effects?
No. Many side effects are on-target-they happen because the drug is doing its job too well in a healthy tissue. For example, diarrhea from metformin or skin rash from EGFR inhibitors are both on-target effects. Off-target effects are when the drug interacts with something completely unrelated to its intended target.
Can off-target effects ever be good?
Yes. Thalidomide was pulled from the market for causing birth defects, but later found to be effective against multiple myeloma due to its off-target immune effects. Sildenafil was originally an angina drug, but its off-target effect on penile blood vessels led to Viagra. Many modern drugs were discovered this way-by noticing unexpected benefits during testing.
Why do some people get side effects and others don’t?
Genetics, age, liver and kidney function, gut bacteria, and even diet can change how a drug behaves in your body. Two people on the same dose might have different off-target interactions because their proteins or enzymes vary. That’s why personalized medicine is growing-because we’re learning that side effects aren’t one-size-fits-all.
Do biologics have fewer side effects than small molecule drugs?
Generally, yes. Biologics like monoclonal antibodies (e.g., Herceptin) are larger, more specific molecules that bind tightly to one target, so they have fewer off-target interactions. Small molecule drugs (like pills) are smaller and more likely to slip into unintended proteins. On average, small molecules have 6.3 off-target interactions; biologics have only 1.2.
How do scientists test for off-target effects?
They use tools like chemical proteomics (to see which proteins a drug binds to), transcriptomics (to see how gene expression changes), and AI models trained on thousands of drug interactions. Companies like Genentech use KinomeScan to test a drug against hundreds of kinases at once. The goal is to catch risky interactions before human trials.

Emma Deasy
March 12, 2026 AT 13:05Let me just say-this is the most comprehensive, meticulously researched breakdown of on-target vs. off-target effects I’ve ever encountered. I’m not exaggerating when I say this article changed how I view every prescription I’ve ever taken. The statins example? Jaw-dropping. The fact that muscle pain isn’t a ‘side effect’ but a *predictable, on-target consequence*? That’s not just science-it’s philosophy. And the part about metformin causing diarrhea because it’s working *too well* in the gut? I’ve been blaming my ‘sensitive stomach’ for years. Turns out, it was just biology being brutally honest.
Also-can we talk about how terrifyingly beautiful it is that thalidomide’s horror became a lifesaver? That’s not coincidence. That’s the universe whispering: ‘Everything is connected.’ I’m tearing up. In a good way. And yes, I cried reading the Herceptin paragraph. No shame.
tamilan Nadar
March 13, 2026 AT 06:02Adam M
March 14, 2026 AT 22:56Rosemary Chude-Sokei
March 15, 2026 AT 10:10I appreciate how thoughtfully this was framed-not just as a pharmacology lesson, but as a reflection on the complexity of human biology. It’s humbling to realize that even our most precise interventions are still blunt instruments in a system as intricate as the human body. I’ve been a clinical researcher for over a decade, and this article reminded me why I chose this field: not to cure everything, but to understand why things go wrong-and sometimes, why they go right in ways we never expected.
The data on biologics vs. small molecules is especially compelling. I’d love to see a follow-up on how polypharmacy interacts with off-target binding. That’s where the real clinical chaos happens.
Noluthando Devour Mamabolo
March 16, 2026 AT 06:15Leah Dobbin
March 17, 2026 AT 11:56How convenient that Big Pharma now pretends to care about ‘off-target effects’-after decades of pushing drugs through trials with minimal screening. The fact that 65% of Phase II failures are due to off-target toxicity? That’s not science. That’s negligence dressed up as innovation. And now they’re patting themselves on the back for using AI to catch what they should’ve caught with basic chemistry in the 1980s?
Don’t mistake improved screening for ethical progress. The same companies that buried thalidomide’s early warnings are now marketing ‘precision medicine’ as if they invented empathy. The real breakthrough isn’t in the lab-it’s in the courtroom.
Ali Hughey
March 18, 2026 AT 12:57WAIT. So you’re telling me the government and pharmaceutical companies HAVE BEEN HIDING this? That side effects aren’t accidents-but predictable outcomes of corporate greed? That’s why my uncle died on statins. That’s why my sister lost her vision on MEK inhibitors. This isn’t science. This is a cover-up. The FDA doesn’t regulate off-target binding because they’re paid by the same labs that design these drugs. KinomeScan? That’s just a PR tool. They don’t test against *all* proteins-only the ones they’re allowed to. There’s a database of 300+ known off-target interactions that never made it to public records. I’ve seen the redacted reports. I have screenshots. I can send them. This is bigger than cancer. This is about control. And they’re using ‘personalized medicine’ as a distraction. Wake up.
Also-why does no one talk about how glyphosate and EMF exposure amplify off-target binding? It’s all connected. The 6.3 average? That’s a conservative estimate. It’s more like 12+ in real-world conditions. I’ve been documenting this since 2016. Nobody listens. But now… now someone finally said it out loud.
Alex MC
March 20, 2026 AT 12:23rakesh sabharwal
March 21, 2026 AT 04:45Aaron Leib
March 22, 2026 AT 07:56Dylan Patrick
March 23, 2026 AT 17:32